 
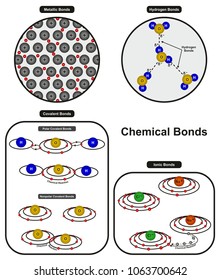
These atoms will share sufficient numbers of electrons in order to achieve a noble gas electron configuration (that is, eight valence electrons).This sharing of valence electrons is called the covalent bond. When two nonmetals bond, they often share electrons since they have similar attractions for them.When two nonmetals bond they often share electrons? A polar bond is a covalent bond between two atoms where the electrons forming the bond are unequally distributed. What are some examples of molecules with polar bonds?Įxamples of molecules with polar bonds include water, hydrogen fluoride, sulfur dioxide, and ammonia. Polar covalent bonds form between atoms with an electronegativity difference between 0.4 and 1.7. Just remember a polar bond refers to a type of covalent bond where electrons aren’t equally shared and electronegativity values are slightly different. What is the electronegativity of a polar bond? When the two atoms are of different elements, the bonding electrons need not be shared equally, resulting in a “polar” bond.When the atoms are alike, as in the H-H bond of H2, the bonding electrons are shared equally (a nonpolar covalent bond).What is the difference between polar and nonpolar covalent bonds? Typically, when pure metal atoms bond together, they prefer metallic bond. However, it is not the way you are talking about. It is very common in transition metal like platinum, palladium. Which of the following is a polar covalent bond? The electronegativity value of phosphorous is 2.1 and the electronegativity value of chlorine is 3.0. Which one of the following is a polar covalent bond? In polar covalent bonds, the electrons are shared unequally, as one atom exerts a stronger force of attraction on the electrons than the other. In pure covalent bonds, the electrons are shared equally. How does a polar covalent bond differ from a covalent bond?Ĭovalent bonds form when electrons are shared between atoms and are attracted by the nuclei of both atoms. Relatively high energies are required to break them (50 – 200 kcal/mol)….Covalent Bonds vs Ionic Bonds. Covalent bonds consist of pairs of electrons shared by two atoms, and bind the atoms in a fixed orientation. They differ in their structure and properties. When it is large, the bond is polar covalent or ionic.Ĭan a covalent bond be between metal and metal? When the difference is very small or zero, the bond is covalent and nonpolar. Whether a bond is ionic, nonpolar covalent, or polar covalent can be estimated by by calculating the absolute value of the difference in electronegativity (ΔEN) of two bonded atoms. How do you know if two elements will form a polar covalent bond? The unequal sharing of the bonding pair results in a partial negative charge on the chlorine atom and a partial positive charge on the hydrogen atom. How do polar covalent bonds occur?Ī polar covalent bond exists when atoms with different electronegativities share electrons in a covalent bond. 
This occurs when one atom has a higher electronegativity than the atom it is sharing with.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
